Photo credits: ScenTree SAS
Do you sell any of the raw materials? Would you like to let our users know?
Send an email to fournisseurs@scentree.coto learn about our advertising opportunities.
Do you sell any of the raw materials? Would you like to let our users know?
Send an email to fournisseurs@scentree.coto learn about our advertising opportunities.
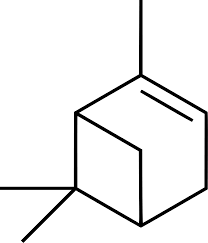
General Presentation
-
CAS N° : 80-56-8
-
EINECS number : 201-291-9
-
FEMA number : 2902
-
FLAVIS number : 01.004
-
JECFA number : 1329
-
Appearance : Colorless liquid
-
Density : 0,858
-
Volatility : Head
-
Price Range : €
Physico-chemical properties
-
Molecular formula : C10H16
-
Molecular Weight : 136,23 g/mol
-
Log P : 4,49
-
Fusion Point : -64°C
-
Boiling Point : 156°C
-
Detection Threshold : 2,5 à 62 ppb selon les personnes (contre 140 ppb pour le Beta-pinène)
-
Optical rotation : Donnée indisponible
-
Vapor pressure : Donnée indisponible
-
Refractive Index @20°C : Donnée indisponible
-
Acid Value : Donnée indisponible.
-
Flash Point : 31°C
Uses
Uses in perfumery :
Alpha-Pinene is used in citrus, fruity and coniferous notes for a fresh touch. Allows to increase the neutrality of a natural raw material. Present in most of citrus fruits and conifers.
Year of discovery :
Data not available.
Natural availability :
Alpha-Pinene is mainly obtained by natural means, by fractional distillation of Turpentine EO of which it is the major compound by far.
Isomerism :
Alpha-Pinene is more camphorated and less woody than beta-Pinene. D-Limonene, Myrcene, Ocimene and Terpinene are constitutional isomers of Pinene. They are also terpenes but have very different olfactory characteristics. The common point of terpenes is that they can be synthesized from Isoprene by a Diels-Alder reaction.
Synthesis precursor :
Alpha-Pinene is a precursor to the synthesis of many compounds of olfactory interest. A hydrogenation of Pinene converts it into Pinane which is a precursor to several compounds used in perfumery. It can be isomerized to beta-Pinene with a very good yield, to proceed to the synthesis of other compounds. It also allows to synthesize Terpineol by hydration. Finally, it allows to synthesize Ocimene by pyrolysis.
Synthesis route :
Alpha-Pinene is a compound mainly obtained with natural means: by fractional distillation of essential oils containing a large amount of it.
Regulations & IFRA
Allergens :
This ingredient does not contain any allergen.
IFRA 51th :
This ingredient is not restricted for the 51th amendment


