Photo credits: ScenTree SAS
Do you sell any of the raw materials? Would you like to let our users know?
Send an email to fournisseurs@scentree.coto learn about our advertising opportunities.
Do you sell any of the raw materials? Would you like to let our users know?
Send an email to fournisseurs@scentree.coto learn about our advertising opportunities.
General Presentation
-
CAS N° :
313973-37-4 -
EINECS number :
441-580-9 -
FEMA number :
Donnée indisponible. -
FLAVIS number :
Donnée indisponible.
-
JECFA number :
Donnée indisponible. -
Volatility :
Head/Heart -
Price Range :
€€€€€
Physico-chemical properties
-
Appearance :
Colorless liquid -
Density :
1,01 -
Refractive Index @20°C :
Donnée indisponible. -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
Donnée indisponible. -
Flash Point :
> 93°C (> 199,4°F)
-
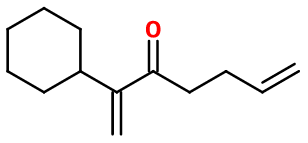
Molecular formula :
C13H20O -
Molecular Weight :
192,3 g/mol -
Log P :
3,5 -
Fusion Point :
Donnée indisponible. -
Boiling Point :
-
Detection Threshold :
Donnée indisponible.
Chemistry & Uses
Uses in perfumery :
Pharaone® is useful as a green booster from the top note, with a fruity effect.
Year of discovery :
1999
Natural availability :
Pharaone® does not exist on a natural state. Thus, it can't be used as extracted from a plant.
Isomerism :
Pharaone® is a constitutional isomer of Alpha-Ionone, Beta-Ionone, Alpha-Damascone® and Beta-Damascone®, but has a much fruitier smell, close to pineapple.
Synthesis precursor :
Pharaone® is not used for the synthesis of another molecule of olfactive interest.
Synthesis route :
The Pharaone® synthesis involves a few steps, starting from Cyclohexyl Acetaldehyde. The first step is a Mannich reaction involving formaldehyde in an acidic medium on the basic reagent, to add an alpha-positioned insaturated ramification, according to its cycle. The second step is a Grignard reaction, between the obtained intermediate product and an organomagnesian as butenylmagnesium bromide, to convert the aldehyde into an alcohol and add a carbon chain to the molecule. The last step is an oxydation that can be carried out using manganese oxide, converting the alcohol into a ketone. This step leads to Pharaone®.
Stability :
Stable in perfumes and in diverse functional bases, except in very acidic bases (detergents) and strong alkaline bases (liquid bleach).
Other comments :
Synergy with Stemone® in a 1:9 ratio of use, to create a tomato leaf effect.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is not restricted for the 51th amendment

















