Photo credits: ScenTree SAS
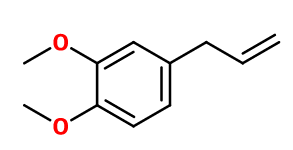
Methyleugenol
Spicy > Cool Spices > Mossy > Earthy
1,2-dimethoxy-4-prop-2-enylbenzene ; 4-allyl veratrole ; 4-allyl-1,2-dimethoxybenzene ; 4-allyl-1,2-dimethoxybenzol ; 4-allylveratrole ; 1,2-dimethoxy-4-(2-propen-1-yl)benzene ; 1,2- dimethoxy-4-allyl benzene ; Eugenol methyl ether ; Veratrole methyl ether ; 1,2-dimethoxy-4-prop-2-enylbenzene

Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
Méthyl Eugenol - 30gr | - |
Visit website
|
- | - | - | |
|
|
Methyl Eugenol | CL-801 |
Visit website
|
Natural |



|
400 Kgs | 100 |
|
|
METHYL EUGENOL | MEG-1 |
Visit website
|
- | 10 grs | 98 |
General Presentation
-
CAS N° :
93-15-2 -
EINECS number :
202-223-0 -
FEMA number :
2475 -
FLAVIS number :
Donnée indisponible.
-
JECFA number :
1790 -
Volatility :
Heart -
Price Range :
€€
Physico-chemical properties
-
Appearance :
Colorless liquid -
Density :
1,035 -
Refractive Index @20°C :
1,532 – 1,536 -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
< 0,075 mmHg @20°C -
Flash Point :
117°C (242,6°F)
-
Molecular formula :
C11H14O2 -
Molecular Weight :
178,23 g/mol -
Log P :
2,4 -
Fusion Point :
-4°C (24,8°F) -
Boiling Point :
248°C (478,4°F) -
Detection Threshold :
68 ppb à 8,5 ppm (0,00085%)
Chemistry & Uses
Uses in perfumery :
Methyl Eugenol is used in rosy floral notes, carnation, lilac, jasmine, hyacinth and other heady flowers. Useful in a chocolate or coffee note.
Year of discovery :
Data not available.
Natural availability :
Methyl Eugenol is present in the Bay St-Thomas EO, Tuberose Absolute and Angelica Root EO among others, Methyl Eugenol can be extracted from these plants in its natural state.
Isomerism :
Methyl Isoeugenol is a position isomer of Methyl Eugenol. Their smell is different : Methyl Isoeugenol is more floral, reminiscent of Tuberose Absolute, while Methyl Eugenol is more spicy. Both are keeping an earthy and wet facet, more noticeable for Methyl Eugenol. Canthoxal® is an isomer of Methyl Eugenol. Its smell is however radically different, as it is marine and aldehydic.
Synthesis precursor :
Methyl Eugenol is not a precursor to the synthesis of another compound of olfactory interest.
Synthesis route :
Methyl Eugenol is obtained synthetically from Eugenol by a methylation reaction. This etherification reaction is made by a Williamson synthesis, which consists in forming a sodium alkoxide in the presence of pure sodium in the reaction medium, and then reacting the obtained eugenolate with methyl chloride.
Stability :
Becomes red under the effect of light. This raw material is not convenient in every functional base : can't be used in a candle or shower gel base.
Other comments :
Methyl Eugenol is one of the 26 allergens in perfumery.
It has a less floral smell than Methyl Isoeugenol.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is restricted by the 51th amendment
- Quantitative limit on the use :
-
Cat.1 Cat.2 Cat.3 Cat.4 Cat.5A B C DCat.6 0,00042 % 0,0015 % 0,00042 % 0,011 % 0,0015 % 0,00021 % 0,00042 % 0,000069 %Cat.5A B C DCat.6 0,0015 % 0,00021 % 0,00042 % 0,000069 %0,0010 % Cat.7A BCat.8 Cat.9 Cat.10A BCat.11A BCat.12 0,00042 % 0,00042 %0,000069 % 0,0017 % 0,00062 % 0,0021 %0,000069 % 0,000069 %Cat.10A BCat.11A BCat.12 0,00062 % 0,0021 %0,000069 % 0,000069 %0,066 %
Annexe I :
Some regulated synthetic ingredients are found in nature and in certain proportions in natural ingredients. This presence in nature has to be taken into account when calculating limits of use recommended by the IFRA. In case you do not know these concentrations, you can use the ones estimated by the IFRA. Here they are :
| List of regulated compounds contained in this ingredient | |||
|---|---|---|---|
| Ingredient Name | Botanical Name | CAS N° | Estimated Concentration |
| Basil oil, chemotype estragole | Ocimum basilicum L. | 8015-73-4 | 0,39 |
| Basil oil, chemotype linalool | Ocimum basilicum L. | 8015-73-4 | 0,2 |
| Basil oleoresin, chemotype estragole | Ocimum basilicum L. | 8015-73-4 | 0,2 |
| Bay leaf, West Indian, oil | Pimenta racemosa (Mill.) J.W. Moore | 8006-78-8 | 2 |
| Calamus oil | Acorus calamus L. | 8015-79-0 | 0,3 |
| Cananga oil | Cananga odorata (Lam.) Hook. f. & Thomson (forma macrophylla Steenis) | 68606-83-7 | 0,2 |
| Cinnamon leaf oil | Cinnamomum zeylanicum Blume | 8015-91-6 | 0,01 |
| Michelia alba flower oil | Michelia x alba DC. (champaca x montana) | 2,06 | |
| Michelia alba leaf oil | Michelia x alba DC. (champaca x montana) | 0,33 | |
| Champaca absolute | Michelia champaca L. | 8006-76-6 | 0,01 |
| Labdanum oil | Cistus ladaniferus L. | 8016-26-0 | 0,05 |
| Citronella oil, Java type | Cymbopogon winterianus Jowitt | 8000-29-1 | 0,1 |
| Clove bud oil | Syzygium aromaticum L. | 8000-34-8 | 0,1 |
| Clove bud oil terpenes | Syzygium aromaticum (L.) Merr. & L.M.Perry | 8000-34-8 | 0,1 |
| Clove leaf oil | Syzygium aromaticum L. | 8000-34-8 | 0,1 |
| Clove stem oil | Syzygium aromaticum L. | 8000-34-8 | 0,06 |
| Elemi oil | Canarium luzonicum (Blume) A. Gray | 8023-89-0 | 0,4 |
| Elemi resinoid | Canarium luzonicum (Blume) A. Gray | 8023-89-0 | 0,09 |
| Tarragon oil | Artemisia dracunculus L. | 8016-88-4 | 0,3 |
| Hyssop oil | Hyssopus officinalis L. | 8006-83-5 | 0,2 |
| Laurel leaf oil | Laurus nobilis L | 8007-48-5 | 3,22 |
| Lemongrass oil, East Indian | Cymbopogon flexuosus (Nees ex Steudel) Will. Watson | 8007-02-1 | 0,05 |
| Mastic absolute | Pistacia lentiscus L. | 68991-39-9 | 0,01 |
| Mastic oil | Pistacia lentiscus L. | 68991-39-9 | 0,02 |
| Lovage root oil | Levisticum officinale Koch | 8016-31-7 | 0,13 |
| Mace oil | Myristica fragrans Houtt. | 8007-12-3 | 1,2 |
| Mace oleoresin | Myristica fragrans Houtt. | 8007-12-3 | 0,6 |
| Myrtle oil | Myrtus communis L. | 8008-46-6 | 1 |
| Nutmeg oil | Myristica fragrans Houtt. | 8008-45-5 | 1,2 |
| Nutmeg oleoresin | Myristica fragrans Houtt. | 8008-45-5 | 0,6 |
| Allspice oil | Pimenta dioica (L.) Merr. | 8006-77-7 | 8,5 |
| Allspice oleoresin | Pimenta officinalis Lindl. | 8006-77-7 | 5,1 |
| Pimenta leaf oil | Pimenta officinalis Lindl. | 8006-77-7 | 6 |
| Rose oil | Rosa x damascena Mill. | 8007-01-0 | 2 |
| Rose water stronger | Rosa x centifolia L. | 8007-01-0 | 0,04 |
| Rose absolute | Rosa x damascena Mill. | 90106-38-0 | 0,43 |
| Rose concrete | Rosa x damascena Mill. | 90106-38-0 | 0,5 |
| Savory winter oil | Satureja montana L. | 0,13 | |
| Tea tree oil | Melaleuca alternifolia (Maiden & Betche) Cheel | 68647-73-4 | 0,05 |
| Thyme absolute | Thymus vulgaris L. | 8007-46-3 | 0,01 |
| Thyme oil, red | Thymus vulgaris L. | 8007-46-3 | 0,01 |
| Thyme oil, white | Thymus vulgaris L. | 8007-46-3 | 0,03 |
| Tuberose absolute | Poliantes tuberosa L. | 8024-05-3 | 1,8 |
| Tuberose concrete | Poliantes tuberosa L. | 8024-05-3 | 1,07 |
| Verbena absolute | Lippia citriodora (L.) Kunth | 8024-12-2 | 0,1 |
| Ylang ylang oil I | Cananga odorata (Lam.) Hook. f. &Thomson oil (forma genuine Steenis) | 8006-81-3 | 0,04 |
| Ylang ylang oil II | Cananga odorata (Lam.) Hook.f.&Thomson forma genuina | 8006-81-3 | 0,03 |
| Ylang ylang oil III | Cananga odorata (Lam.) Hook. f. &Thomson oil (forma genuine Steenis) | 8006-81-3 | 0,04 |
| Bucchu absolute, betulina | Agathosma betulina (P.J.Bergius) Pillans | 84649-93-4 | 0,02 |
| Bucchu oil, crenulata | Agathosma crenulata (L.) Pill. | 92346-82-2 | 0,19 |
| Hyacinth absolute | Hyacinthus orientalis L. | 8023-94-7 | 1,5 |
| Ocatea cymbarum oil | Ocotea cymbarum Kunth | 68917-09-9 | 0,3 |
| Sassafras bark oil | Sassafras albidum (Nutt.) Nees | 8006-80-2 | 0,3 |
| Snakeroot oil | Asarum canadense L. | 8016-69-1 | 40 |



















