Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
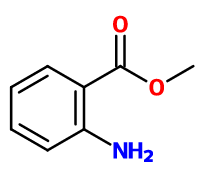
Methyl Anthranilate | - |
Visit website
|
Synthetic Aroma Chemicals | - | - | |
|
|
Anthranilate de Methyle - 30 Gr | - |
Visit website
|
- | - | - | |
|
|
METHYL ANTHRANILATE 10KG DRUM STEEL | 442FP24223 |
Visit website
|
Synthetic Aroma Chemicals |

|
- | - |
|
|
METHYL ANTHRANILATE - A- TOCOPHEROL (AROMES) | AN-1 |
Visit website
|
- |


|
10 grs | - |
General Presentation
-
CAS N° :
134-20-3 -
EINECS number :
205-132-4 -
FEMA number :
2682 -
FLAVIS number :
09.715
-
JECFA number :
1534 -
Volatility :
Heart -
Price Range :
€
Physico-chemical properties
-
Appearance :
Colorless liquid to solid -
Density :
1,166 -
Refractive Index @20°C :
1,581 – 1,585 -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
0,0097 mmHg @20°C 0,0157 mmHg @25°C -
Flash Point :
123°C (253,4°F)
-
Molecular formula :
C8H9NO2 -
Molecular Weight :
151,16 g/mol -
Log P :
1,66 -
Fusion Point :
24°C (75,2°F) -
Boiling Point :
255°C (491°F) -
Detection Threshold :
0,12 ng/l air
Chemistry & Uses
Uses in perfumery :
Methyl Anthranilate is used for wild strawberry and wild fruit notes, in orange blossom and exotic flower accords: gardenia, tuberose, jasmine.
Year of discovery :
1898
Natural availability :
Methyl Anthranilate is present in Grandiflorum Jasmine Absolute, Neroli EO, Ylang-Ylang Extra EO (and other ylang fractions), Champaca Absolute, in grapes and citrus fruits, from which it can be extracted in its natural state.
Isomerism :
Methyl Anthranilate does not have any isomer used in perfumery.
Synthesis precursor :
Methyl Anthranilate has the ability to easily react with aldehydes to form molecules called Schiff bases, which can serve as a perfumery ingredient or base for a Maillard reaction. For example, Aurantiol (Schiff Base) results from the reaction of this molecule with Hydroxycitronellal and is a raw material also used in perfumery. The Schiff base with Lilial® (Verdantiol) or the one with Triplal® (Vertosine) are also much used.
Synthesis route :
Methyl Anthranilate is synthesized either by esterification of anthranilic acid with methanol or by reaction between methanol and isatoic anhydride, releasing CO2.
Stability :
Methyl Anthranilate is well known for synthesizing Schiff bases by reacting espacially with aldehydes. These compounds can have an olfactive interest but are still a source of coloration in perfume concentrates and perfumes besides others. This is why this raw material is used is small quantities.
Exclusively stable in fabric conditioners, shampoos and hair conditioners
Other comments :
Methyl Anthranilate is more associated with Orange Blossom Absolute notes and fruity notes of wild strawberry than Dimethyl Anthranilate, closer to Mandarin Yellow EO.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is not restricted for the 51th amendment



















