Photo credits: ScenTree SAS
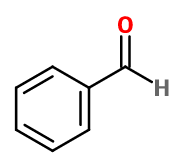
Benzaldehyde
Balsamic Ambery > Almondy > Vanillic
Benzoic Aldehyde ; Almond artificial oil ; Benzanoaldehyde ; Benzarone ; Benzene carbaldehyde ; Benzene carbonal ; Benzene carboxaldehyde ; Benzene methylal ; Benzene carbinal ; Benzenecarbonal ; Benzenemehylal ; Benzoyl hydride ; Phenyl methanal ; Phenylformaldehyde ; Phenylketone ; Phenylmethanal

Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
Benzaldehyde natural | - |
Visit website
|
Natural Aroma Chemicals | - | - | |
|
|
Benzaldehyde Synthetic | - |
Visit website
|
Synthetic Aroma Chemicals | - | - | |
|
|
Benzaldéhyde - 30gr | - |
Visit website
|
- | - | - | |
|
|
BENZALDEHYDE | ALDB-1 |
Visit website
|
- | 10 grs | - |
General Presentation
-
CAS N° :
100-52-7 -
EINECS number :
202-860-4 -
FEMA number :
2127 -
FLAVIS number :
05.013
-
JECFA number :
22 -
Volatility :
Head -
Price Range :
€
Physico-chemical properties
-
Appearance :
Colorless viscous liquid -
Density :
1,045 -
Refractive Index @20°C :
1,544 - 1,547 -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
1,2676 mmHg @25°C 3,7503 mmHg @45°C -
Flash Point :
64°C (147,2°F)
-
Molecular formula :
C7H6O -
Molecular Weight :
106,12 g/mol -
Log P :
1,5 -
Fusion Point :
-26°C (-14,8°F) -
Boiling Point :
179°C (354,2°F) -
Detection Threshold :
Varie selon les personnes entre 100 ppb et 4,6 ppm (0,00046%), lorsque son seuil de reconnaissance varie entre 330 ppb et 4,1 ppm
Chemistry & Uses
Uses in perfumery :
Benzaldehyde is used in almond, fruit stone and nutty notes. Gives nuances to vanilla notes.
Year of discovery :
Discovered in 1863.
Natural availability :
Benzaldehyde can be found in almond, several fruits, Vanilla Bourbon Absolute (and other origins), Grandiflorum Jasmine Absolute and fruit stones. In its natural state, it is extracted from Bitter Almond EO, of which it is the major compound.
Isomerism :
Benzaldehyde does not have any isomer used in perfumery.
Synthesis precursor :
Benzaldehyde is the precursor to the synthesis of many compounds used in perfumery. For example, its hydrogenation synthesizes benzoic alcohol. It condenses with other aldehydes to create many other compounds, replacing the aldehyde function with an alcene function. Finally, a Perkin reaction with several acids allows to obtain various compounds, such as Cinnamic Acid by reaction with acetic anhydride. Finally, benzaldehyde forms a Schiff base by reaction with Methyl Anthranilate or Indole for example.
Synthesis route :
Benzaldehyde is synthesized by hydrolysis of benzyl chloride, by an acidic route in the presence of iron chloride, or by an alkaline route in the presence of sodium carbonate. A toluene oxidation also allows to obtain both Benzoic Acid and Benzaldehyde.
Stability :
Tends to oxydize easily into perbenzoic acid, which can react with itself to onvert it into Benzoic Acid.
Tends to color through time.
Other comments :
As it can be compared to Furfural and Salicylaldehyde, Benzaldehyde is the reference almond note in perfumery.
Labelling
Allergens :
This ingredient is classified as an allergen under European Regulation 2023/1545, dated August 26, 2023.
Its presence must therefore be declared on product labels when it exceeds 0.001% in leave-on products and 0.01% in rinse-off products.
IFRA
IFRA 51th :
This ingredient is restricted by the 51th amendment
- Quantitative limit on the use :
-
Cat.1 Cat.2 Cat.3 Cat.4 Cat.5A B C DCat.6 0,045 % 0,014 % 0,27 % 0,25 % 0,064 % 0,064 % 0,064 % 0,021 %Cat.5A B C DCat.6 0,064 % 0,064 % 0,064 % 0,021 %0,15 % Cat.7A BCat.8 Cat.9 Cat.10A BCat.11A BCat.12 0,52 % 0,52 %0,021 % 0,49 % 0,49 % 1,8 %0,021 % 0,021 %Cat.10A BCat.11A BCat.12 0,49 % 1,8 %0,021 % 0,021 %No Restriction
Annexe I :
Some regulated synthetic ingredients are found in nature and in certain proportions in natural ingredients. This presence in nature has to be taken into account when calculating limits of use recommended by the IFRA. In case you do not know these concentrations, you can use the ones estimated by the IFRA. Here they are :
| List of regulated compounds contained in this ingredient | |||
|---|---|---|---|
| Ingredient Name | Botanical Name | CAS N° | Estimated Concentration |
| Almond oil, bitter | Prunus amygdalus amara (Bitter Almond) kernel oil | 8013-76-1 | 99 |
| Benzoin infusion, Sumatra | Styrax paralleloneurum Perkins | 0,001 | |
| Cajuput oil | Melaleuca leucadendron L. | 8008-98-8 | 0,1 |
| Cinnamon bark oil, Laos | Cinnamomum loureiroi Nees | 97659-68-2 | 0,5 |
| Cinnamon bark CO2 extract | Cinnamomum verum J. Presl syn. C. zeylanicum Blume | 8015-91-6 | 0,1 |
| Cinnamon bark oil | Cinnamomum zeylanicum Blume | 8015-91-6 | 0,2 |
| Cinnamon leaf oil | Cinnamomum zeylanicum Blume | 8015-91-6 | 0,16 |
| Cassia bark oil | Cinnamomum cassia [L.] J. Presl syn. C. aromaticum Nees | 8007-80-5 | 1,3 |
| Cassia bark oleoresin | Cinnamomum cassia [L.] J. Presl syn. C. aromaticum Nees | 8007-80-5 | 0,95 |
| Cassia oil | Cinnamomum cassia [L.] J. Presl syn. C. aromaticum Nees | 8007-80-5 | 1 |
| Cassie absolute | Vachellia farnesiana (L.) Willd. | 8023-82-3 | 0,3 |
| Cistus absolute | Cistus ladaniferus L. | 8016-26-0 | 0,6 |
| Cistus concrete | Cistus ladaniferus L. | 8016-26-0 | 0,6 |
| Cistus oil | Cistus ladaniferus L. | 8016-26-0 | 0,9 |
| Labdanum oil | Cistus ladaniferus L. | 8016-26-0 | 0,38 |
| Labdanum oleoresin | Cistus ladaniferus L. | 8016-26-0 | 0,2 |
| Davana oil | Artemisia pallens Wall. | 8016-03-3 | 0,2 |
| Jasmine grandiflorum absolute | Jasminum grandiflorum L. | 8022-96-6 | 0,02 |
| Mimosa absolute | Acacia decurrens (Wendl.f.) Willd. | 8031-03-6 | 0,08 |
| Niaouli oil | Melaleuca viridiflora Sol. ex Gaertn. | 8014-68-4 | 0,2 |
| Perilla oil | Perilla frutescens (L.) Britton | 68132-21-8 | 0,33 |
| Rose oil | Rosa x damascena Mill. | 8007-01-0 | 0,04 |
| Rose absolute | Rosa x damascena Mill. | 90106-38-0 | 0,02 |
| Rose concrete | Rosa x damascena Mill. | 90106-38-0 | 0,5 |
| Styrax extract | Liquidambar spp. | 8046-19-3 | 0,05 |
| Styrax oil | Liquidambar styraciflua L. | 8046-19-3 | 0,03 |
| Styrax oil, Honduras | Liquidambar styraciflua L. | 8046-19-3 | 0,1 |
| Styrax resinoid | Liquidambar styraciflua L. | 8046-19-3 | 0,04 |
| Styrax oil, pyrogenated | Liquidambar orientalis Mill. | 8024-01-9 | 0,1 |
| Hyacinth absolute | Hyacinthus orientalis L. | 8023-94-7 | 0,1 |
| Labdanum extract ambreine | Cistus ladaniferus L. | 68917-77-1 | 0,8 |
| Labdanum gum | Cistus labdanifer L. | 8016-26-0; 89997-74-0, 84775-64-4 | 0,2 |
| Labdanum resinoid | Cistus ladaniferus L. | 89997-74-0 | 0,03 |



















