Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
SCATOLE 1% IPM | SC1IPM-1 |
Visit website
|
- | 10 grs | - |
General Presentation
-
CAS N° :
83-34-1 -
EINECS number :
201-471-7 -
FEMA number :
3019 -
FLAVIS number :
14.004
-
JECFA number :
1304 -
Volatility :
Base -
Price Range :
€€€€
Physico-chemical properties
-
Appearance :
White solid -
Density :
Solide -
Refractive Index @20°C :
Donnée indisponible. -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
Donnée indisponible. -
Flash Point :
132°C (269,6°F)
-
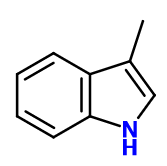
Molecular formula :
C9H9N -
Molecular Weight :
131,17 g/mol -
Log P :
2,60 -
Fusion Point :
96°C (204,8°F) -
Boiling Point :
266°C (510,8°F) -
Detection Threshold :
0,2 ppb (0,00000002%)
Chemistry & Uses
Uses in perfumery :
Skatole is used in small quantities in leather, woody notes, but less in floral notes than Indole. Brings an animalic facet to all types of accords.
Year of discovery :
1880
Natural availability :
Skatole is found naturally in some cheeses, among others, but is very little extracted for its use in perfumery. Synthetic skatole is the most used.
Isomerism :
Skatole does not have isomer used in perfumery.
Synthesis precursor :
Skatole is not a precursor to the synthesis of another compound of olfactory interest.
Synthesis route :
Skatole is part of the same family of molecules as Indole: the pyroles. These molecules can be synthesized in several ways. In the case of Skatole, a Fischer synthesis is adapted. This reaction consists in reacting phenylhydrazine with propanal, catalysed by a strong acid. Many other synthetic routes exist for this compound, such as the synthesis of Bischler-Möhlau, the Reissert or the Madelung synthesis among others.
Stability :
This compound is well known for synthesizing Schiff bases by reacting espacially with aldehydes. These compounds can have an olfactive interest but are still a source of coloration in perfume concentrates and perfumes besides others. This is why this raw material is used is small quantities.
Other comments :
Skatole is more powerful and greedy than Indole but less floral.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is not restricted for the 51th amendment



















