Photo credits: ScenTree SAS
Do you sell any of the raw materials? Would you like to let our users know?
Send an email to fournisseurs@scentree.coto learn about our advertising opportunities.
Do you sell any of the raw materials? Would you like to let our users know?
Send an email to fournisseurs@scentree.coto learn about our advertising opportunities.
General Presentation
-
CAS N° :
13257-44-8 -
EINECS number :
236-249-9 -
FEMA number :
Donnée indisponible. -
FLAVIS number :
Donnée indisponible.
-
JECFA number :
Donnée indisponible. -
Volatility :
Heart/Base -
Price Range :
€€€€
Physico-chemical properties
-
Appearance :
Colorless liquid -
Density :
0,88 -
Refractive Index @20°C :
1,439 -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
Donnée indisponible. -
Flash Point :
96°C (204,8°F)
-
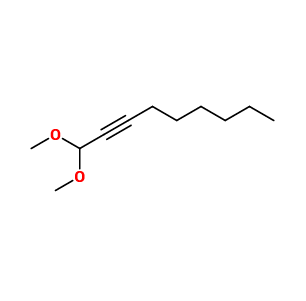
Molecular formula :
C11H20O2 -
Molecular Weight :
184,27 g/mol -
Log P :
2,94 -
Fusion Point :
Donnée indisponible. -
Boiling Point :
228°C (442,4°F) -
Detection Threshold :
Donnée indisponible.
Chemistry & Uses
Uses in perfumery :
Provides a fruity pear/apple facet with watery and floral-violet nuances. Please note, that this compound is IFRA-regulated.
Year of discovery :
Data not available.
Natural availability :
Parmavert® is not found in nature.
Isomerism :
Parmavert® is a constitutional isomer of aldehyde C-14 and Citronellyl Formate. However, its smell is quite different from the latter two compounds.
Synthesis precursor :
Parmavert® is not a precursor to the synthesis of another compound of olfactory interest.
Synthesis route :
Parmavert® can be synthesized by an acetalization reaction of nonynaldehyde (nonynal). This reaction puts the aldehyde in the presence of an acid catalyst followed by methanol. This stage leads to the formation of a hemiacetal. The second step is a nucleophilic addition. In an acidic medium, a new methanol molecule can be added to the intermediate molecule to form Parmavert®. The equilibrium of those two reactions can be shifted by using a Dean-Stark separator.
Stability :
Stable in perfumes and diverse functional bases
Other comments :
Data not available.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is restricted by the 51th amendment
- Quantitative limit on the use :
-
Cat.1 Cat.2 Cat.3 Cat.4 Cat.5A B C DCat.6 1,8 % 0,53 % 11 % 9,9 % 2,5 % 2,5 % 2,5 % 2,5 %Cat.5A B C DCat.6 2,5 % 2,5 % 2,5 % 2,5 %5,8 % Cat.7A BCat.8 Cat.9 Cat.10A BCat.11A BCat.12 20 % 20 %1 % 19 % 69 % 69 %38 % 38 %Cat.10A BCat.11A BCat.12 69 % 69 %38 % 38 %No Restriction


















