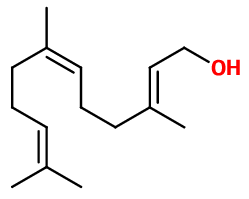
Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
Farnésol - 30gr | - |
Visit website
|
- | - | - | |
|
|
FARNESOL SYNTHETIQUE | FARN-1 |
Visit website
|
- | 10 grs | - |
General Presentation
-
CAS N° :
4602-84-0 -
EINECS number :
225-004-1 -
FEMA number :
2478 -
FLAVIS number :
02.029
-
JECFA number :
1230 -
Volatility :
Base -
Price Range :
€€€
Physico-chemical properties
-
Appearance :
Colorless liquid -
Density :
0,88 -
Refractive Index @20°C :
Donnée indisponible. -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
Donnée indisponible. -
Flash Point :
96°C (204,8°F)
-
Molecular formula :
C15H26O -
Molecular Weight :
222,37 g/mol -
Log P :
5,7 -
Fusion Point :
Donnée indisponible. -
Boiling Point :
107°C (224,6°F) -
Detection Threshold :
1 ppm (0,0001%)
Chemistry & Uses
Uses in perfumery :
Farnesol is used for lily of the valley notes, to bring a fruity nuance. It is also used as a fixative.
Year of discovery :
Discovered in 1913
Natural availability :
Farnesol can be extracted from Ambrette Seeds Absolute, because it is found there in its (Z,E) conformation. It is also found in Petitgrain Bigarade EO and Orange Blossom Absolute, from which it can be extracted.
Isomerism :
Farnesol has four stereoisomers, due to the presence of two double bonds. Thus, these two bonds can have either a cis (Z) or trans (E) configuration. The four stereoisomer couples of Farnesol are : (Z,Z), (E,E), (Z,E) and (E,Z). In perfumery, a mixture of isomers is used most of the time, because no real olfactive difference exist between the isomers.
Synthesis precursor :
Farnesol is not used for the synthesis of another compound of olfactive interest.
Synthesis route :
Synthetic Farnesol can be obtained by isomerization of Nerolidol. This synthesis is mostly replacing the natural extraction of Farnesol, as it was formerly usual.
Stability :
Terpenes may polymerize under strong oxydation.
Other comments :
The smell of Farnesol becomes stronger when the molecule is evaporating, surely due to oxydation.
Farnesol is part of the sesquiterpene molecule category. This means that this molecule has fifteen carbon atoms, that is three isoprene units. Terpenes (monoterpenes, diterpenes, sesquiterpenes...) were discovered thanks to the Diels-Alder reaction, involving a diene and a dienophile, abling to modelize terpenes and to obtain a great variety of them.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is restricted by the 51th amendment
- Quantitative limit on the use :
-
Cat.1 Cat.2 Cat.3 Cat.4 Cat.5A B C DCat.6 0,21 % 0,062 % 1,2 % 1,2 % 0,29 % 0,29 % 0,29 % 0,29 %Cat.5A B C DCat.6 0,29 % 0,29 % 0,29 % 0,29 %0,68 % Cat.7A BCat.8 Cat.9 Cat.10A BCat.11A BCat.12 2,4 % 2,4 %0,12 % 2,3 % 8,1 % 8,1 %4,5 % 4,5 %Cat.10A BCat.11A BCat.12 8,1 % 8,1 %4,5 % 4,5 %No Restriction
Annexe I :
Some regulated synthetic ingredients are found in nature and in certain proportions in natural ingredients. This presence in nature has to be taken into account when calculating limits of use recommended by the IFRA. In case you do not know these concentrations, you can use the ones estimated by the IFRA. Here they are :
| List of regulated compounds contained in this ingredient | |||
|---|---|---|---|
| Ingredient Name | Botanical Name | CAS N° | Estimated Concentration |
| Yarrow oil | Achillea millefolium L. | 8022-07-9 | 0,1 |
| Ambrette seed absolute | Hibiscus abelmoschus L. | 8015-62-1 | 5 |
| Ambrette seed oil | Hibiscus abelmoschus L. | 8015-62-1 | 3 |
| Ambrette tincture | Hibiscus abelmoschus L. | 8015-62-1 | 0,2 |
| Arnica absolute | Arnica montana L. | 8057-65-6 | 1,2 |
| Arnica oils, montana | Arnica montana L. | 8057-65-6 | 5 |
| Schinus molle oil | Schinus areira L. | 68917-52-2 | 0,03 |
| Cabreuva oil | Myrocarpus frondosus Fr. Allem | 68188-03-4 | 0,1 |
| Calamus oil | Acorus calamus L. | 8015-79-0 | 0,5 |
| Cananga oil | Cananga odorata (Lam.) Hook. f. & Thomson (forma macrophylla Steenis) | 68606-83-7 | 2 |
| Cinnamon bark oil, Laos | Cinnamomum loureiroi Nees | 97659-68-2 | 0,01 |
| Cinnamon leaf oil | Cinnamomum zeylanicum Blume | 8015-91-6 | 0,12 |
| Carrot seed oil | Daucus carota L. | 8015-88-1 | 2,18 |
| Citron oil | Citrus medica L. | 68991-25-3 | 0,01 |
| Orange blossoms absolute | Citrus aurantium L. spp. Amara Link | 8016-38-4 | 3,72 |
| Palmarosa oil | Cymbopogon martinii (Roxb.) Wats | 8014-19-5 | 1 |
| Rose absolute | Rosa x damascena Mill. | 90106-38-0 | 0,32 |
| Thyme absolute | Thymus vulgaris L. | 8007-46-3 | 0,02 |
| Tuberose absolute | Poliantes tuberosa L. | 8024-05-3 | 0,3 |
| Tuberose concrete | Poliantes tuberosa L. | 8024-05-3 | 0,3 |
| Ylang, Ylang oil complete | Cananga odorata (Lam.) Hook. f. &Thomson oil (forma genuine Steenis) | 8006-81-3 | 1,5 |
| Ylang, Ylang oil extra | Cananga odorata (Lam.) Hook. f. &Thomson oil (forma genuine Steenis) | 8006-81-3 | 1,36 |
| Baccharis dracunculifolia oil | Baccharis dracunculifolia | 68991-21-9 | 0,2 |
| Orange flower, bitter, concrete | Citrus aurantium L. | 72968-50-4 | 1,95 |
| Orange flower water absolute | Citrus aurantium L. spp. Amara Link | 8030-28-2 | 1,08 |
| Orange flower, bitter, CO2 extract | Citrus aurantium L. | 8030-28-2 | 6,5 |
| Petitgrain bigarade absolute | Citrus aurantium L. | 8030-28-2 | 0,11 |
-
Specified ingredients: notes
Farnesol should only be used as a fragrance ingredient if it contains a minimum of 96% of farnesol isomers as determined by GLC.



















