Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
ETHYL ROSATE | ES-2 |
Visit website
|
- |

|
10 grs | - |
General Presentation
-
CAS N° :
35044-59-8 -
EINECS number :
252-335-9 -
FEMA number :
Donnée indisponible. -
FLAVIS number :
Donnée indisponible.
-
JECFA number :
Donnée indisponible. -
Volatility :
Head/Heart -
Price Range :
€€€
Physico-chemical properties
-
Appearance :
Colorless liquid -
Density :
0,967 -
Refractive Index @20°C :
Donnée indisponible. -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
Donnée indisponible. -
Flash Point :
85°C (185°F)
-
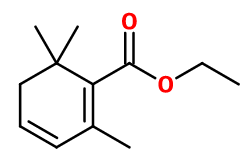
Molecular formula :
C12H18O2 -
Molecular Weight :
194,27 g/mol -
Log P :
Donnée indisponible. -
Fusion Point :
Donnée indisponible. -
Boiling Point :
239°C (462,2°F) -
Detection Threshold :
Donnée indisponible.
Chemistry & Uses
Uses in perfumery :
Ethyl Safranate is used in saffron notes, fruity notes of yellow fruits such as plum and other stone fruits, for notes of cooked apple, in woody perfumes to bring a fruity note and in floral notes. It can be used in various alkaline bases for its stability.
Year of discovery :
1955
Natural availability :
Ethyl Safranate is not available in its natural state.
Isomerism :
The Ethyl Safranate most commonly used in perfumery is a mixture of alpha, beta and gamma isomers of the molecule. These isomers are not isolated for their use in perfumery. In addition, Ethyl Safranate is a constitutional isomer of Scentenal® and Hyacinth Body®. Their smell is however very different.
Synthesis precursor :
Ethyl Safranate does not interfere with the synthesis of another compound of olfactory interest.
Synthesis route :
Ethyl Safranate can be synthesized by a reaction between Mesityl Oxide and Ethyl Acetoacetate. Both will cyclize when put in an acid medium. The obtained intermediate molecule can be reduced and dehydrated to obtain Ethyl Safranate.
Stability :
Esters may form acetic acid through time. Ethyl Safranate still is globally stable in perfumes and diverse functional bases, except acid cleaners and very alkaline products.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is not restricted for the 51th amendment



















