Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
SCENTENAL® | 981810 |
Visit website
|
Molecules |

|
- | - |
|
|
SCENTENAL | SCE-1 |
Visit website
|
- | 10 grs | - |
General Presentation
-
CAS N° :
86803-90-9 -
EINECS number :
429-860-9 -
FEMA number :
Donnée indisponible. -
FLAVIS number :
Donnée indisponible.
-
JECFA number :
Donnée indisponible. -
Volatility :
Base -
Price Range :
€€€
Physico-chemical properties
-
Appearance :
Colorless liquid -
Density :
1,075 -
Refractive Index @20°C :
Donnée indisponible. -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
Donnée indisponible. -
Flash Point :
>100°C (>212°F)
-
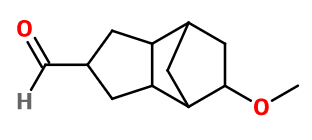
Molecular formula :
C12H18O2 -
Molecular Weight :
194,27 g/mol -
Log P :
Donnée indisponible. -
Fusion Point :
Donnée indisponible. -
Boiling Point :
246°C (474,8°F) -
Detection Threshold :
Donnée indisponible.
Chemistry & Uses
Uses in perfumery :
Scentenal® is used in marine notes, juicy fruits, white and light flowers, violet and orris root notes. Particularly suitable for green notes.
Year of discovery :
Scentenal®' trademark has been published and protected by Firmenich SA since 17/03/1989 (brand N°536294)
Natural availability :
Scentenal® is not available in its natural state.
Isomerism :
Hyacinth Body® is a constitutional isomer of Scentenal®, but it does not have a similar smell at all, as it is floral-rosy and green.
Synthesis precursor :
Scentenal® is not a precursor to the synthesis of another compound of olfactory interest.
Synthesis route :
Data not available.
Stability :
Stable in perfumes and diverse functional bases
Other comments :
In comparision to Helional®, Scentenal® has a more aldehydic smell.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is restricted by the 51th amendment
- Quantitative limit on the use :
-
Cat.1 Cat.2 Cat.3 Cat.4 Cat.5A B C DCat.6 0,19 % 0,057 % 1,2 % 1,1 % 0,27 % 0,27 % 0,27 % 0,091 %Cat.5A B C DCat.6 0,27 % 0,27 % 0,27 % 0,091 %0,63 % Cat.7A BCat.8 Cat.9 Cat.10A BCat.11A BCat.12 2,2 % 2,2 %0,091 % 2,1 % 2,1 % 7,5 %0,091 % 0,091 %Cat.10A BCat.11A BCat.12 2,1 % 7,5 %0,091 % 0,091 %No Restriction



















