Photo credits: ScenTree SAS

Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
PADMA/Phenylacetaldehyde dimethyl acetal | - |
Visit website
|
Synthetic Aroma Chemicals | - | - | |
|
|
Phenylacetaldehyde DMA | 2311172010 |
Visit website
|
Synthetic Aroma Chemicals |

|
- | - |
|
|
PHENYLACETALDEHYDE DIMETHYL ACETAL | PADA-1 |
Visit website
|
- | 10 grs | - |
General Presentation
-
CAS N° :
101-48-4 -
EINECS number :
202-945-6 -
FEMA number :
2876 -
FLAVIS number :
06.006
-
JECFA number :
1003 -
Volatility :
Head/Heart -
Price Range :
€
Physico-chemical properties
-
Appearance :
Colorless liquid -
Density :
1,004 -
Refractive Index @20°C :
Donnée indisponible. -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
Donnée indisponible. -
Flash Point :
88°C (190,4°F)
-
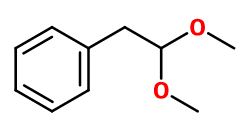
Molecular formula :
C10H14O2 -
Molecular Weight :
166,22 g/mol -
Log P :
1,9 -
Fusion Point :
Donnée indisponible. -
Boiling Point :
220°C (428°F) -
Detection Threshold :
Donnée indisponible.
Chemistry & Uses
Uses in perfumery :
Viridine® is used in rose, hyacinth, honeysuckle, lily of the valley, lilac, carnation, gardenia, lily and geranium notes. Useful in woody-patchouli and vetiver notes. Brings a ripe fruit effect to fruit accords. Also used in eaux fraiches and in fougere perfumes.
Year of discovery :
Data not available.
Natural availability :
Viridine® is not available in its natural state.
Isomerism :
Viridine® is a constitutional isomer of DihydroEugenol, even if both molecules do not have the same smell.
Synthesis precursor :
Viridine® is not a precursor to the synthesis of another compound of olfactory interest.
Synthesis route :
Viridine® is synthesized by an acetalization reaction between Phenylacetic Aldehyde (synthesized from styrene oxide for example) and methanol. This reaction is catalysed by a strong concentrated acid.
Stability :
Stable in perfumes and diverse functional bases
Other comments :
Viridine® has a distinctive mushroom and metallic note from other rosy materials.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is not restricted for the 51th amendment



















