Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
Triathyl Citrate | - |
Visit website
|
Synthetic Aroma Chemicals | - | - | |
|
|
Triéthyl Citrate - 30gr | - |
Visit website
|
- | - | - | |
|
|
TRIETHYL CITRATE | CITET-1 |
Visit website
|
- | 10 grs | 99 - 100 | |
|
|
TRIETHYL CITRATE NATURAL | CITETNAT-1 |
Visit website
|
Naturel | 10 grs | - |
General Presentation
-
CAS N° :
77-93-0 -
EINECS number :
201-070-7 -
FEMA number :
3083 -
FLAVIS number :
09.512
-
JECFA number :
629 -
Volatility :
NON TROUVE_N/A -
Price Range :
€
Physico-chemical properties
-
Appearance :
Colorless liquid -
Density :
1,14 -
Refractive Index @20°C :
Donnée indisponible. -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
Donnée indisponible. -
Flash Point :
155°C (311°F)
-
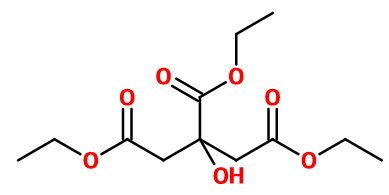
Molecular formula :
C12H20O7 -
Molecular Weight :
276,28 g/mol -
Log P :
1,17 -
Fusion Point :
Donnée indisponible. -
Boiling Point :
294°C (561,2°F) -
Detection Threshold :
Donnée indisponible.
Chemistry & Uses
Uses in perfumery :
Triethylcitrate is a solvent commonly found in perfume concentrates, as it is mainly used to dilute raw materials that are too viscous or pasty to be used as they are.
Year of discovery :
Data not available.
Natural availability :
Triethyl Citrate is naturally present in cabbages and some white wines. Natural Triethyl Citrate is synthesized by hemi-synthesis using natural citric acid (lemon extract, for example) and ethanol.
Isomerism :
Triethyl Citrate does not have any isomer used in perfumery.
Synthesis precursor :
Triethyl Citrate is not a precursor to the synthesis of another compound of olfactory interest.
Synthesis route :
Triethyl Citrate is a triester of citric acid. It is therefore obtained by an extensive esterification of citric acid, by reaction with an excess of ethanol in the presence of an acid catalyst such as concentrated sulfuric acid. The end of the reaction can be evaluated by an acid-base titration: when the acid concentration is stable, the reaction is at equilibrium.
Stability :
Data not available.
Other comments :
Data not available.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is not restricted for the 51th amendment



















