Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
Tetrahydrolinalool | - |
Visit website
|
Synthetic Aroma Chemicals | - | - | |
|
|
Tetrahydrolinalool | 30786547 |
Visit website
|
Molecule | - | - | |
|
|
Tetrahydrolinalool BMBcert™ | 30786547 |
Visit website
|
Molecule | - | - | |
|
|
TETRAHYDROLINALOOL | TETL-1 |
Visit website
|
- | 10 grs | 99.0 - 100.0 |
General Presentation
-
CAS N° :
78-69-3 -
EINECS number :
201-133-9 -
FEMA number :
3060 -
FLAVIS number :
Donnée indisponible.
-
JECFA number :
357 -
Volatility :
Head -
Price Range :
Data not available.
Physico-chemical properties
-
Appearance :
Colorless liquid -
Density :
0,826 -
Refractive Index @20°C :
Donnée indisponible. -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
0,75 mmHg @20°C 2,25 mmHg @50°C -
Flash Point :
77°C (170,6°F)
-
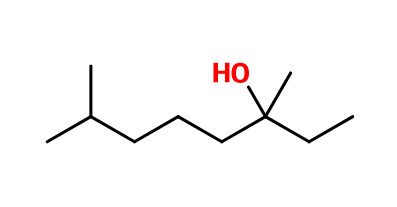
Molecular formula :
C10H22O -
Molecular Weight :
158,28 g/mol -
Log P :
3,3 -
Fusion Point :
197°C (386,6°F) -
Boiling Point :
197°C (386,6°F) -
Detection Threshold :
0,02 μg/l
Chemistry & Uses
Uses in perfumery :
Tetrahydrolinalool is a product used mainly to replace linalool in applications where the latter may not be stable enough. It serves as a more stable alternative to linalool, although it has a fresher and more technical scent.
Year of discovery :
Data not available.
Natural availability :
Tetrahydrlinalool is present in nature.
Isomerism :
Data not available.
Synthesis precursor :
Tetrahydrolinalool is not used to prepare other products used in perfumery
Synthesis route :
May be prepared by hydrogenation of linalool
Stability :
Tetrahydrolinalool is very stable is all kind of commun application, it is more stable than the linalol itself.
Other comments :
Data not available.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is not restricted for the 51th amendment


















