Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
RHUBOFIX® | 978880 |
Visit website
|
Molecules | - | - | |
|
|
Rhubofix® - 30gr | - |
Visit website
|
- | - | - | |
|
|
RHUBOFIX | RHUB-1 |
Visit website
|
- |


|
10 grs | 70.0 - 100.0 |
General Presentation
-
CAS N° :
41816-03-9 -
EINECS number :
255-560-0 -
FEMA number :
Donnée indisponible. -
FLAVIS number :
Donnée indisponible.
-
JECFA number :
Donnée indisponible. -
Volatility :
Base -
Price Range :
€€€
Physico-chemical properties
-
Appearance :
Colorless liquid -
Density :
1,011 -
Refractive Index @20°C :
Donnée indisponible. -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
Donnée indisponible. -
Flash Point :
>100°C (>212°F)
-
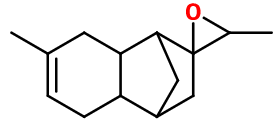
Molecular formula :
C14H20O -
Molecular Weight :
204,31 g/mol -
Log P :
2,45-3,85 -
Fusion Point :
< -20°C (< -4°F) -
Boiling Point :
281°C (537,8°F) -
Detection Threshold :
Donnée indisponible.
Chemistry & Uses
Uses in perfumery :
Rhubofix® is used in rhubarb notes, in a green top note and in grapefruit accords. Allows to keep a green, sulfuric and rhubarb note throughout the evaporation of a fragrance.
Year of discovery :
1972 'Rhubofix®' trademark has been published and protected by Firmenich SA since 22/04/1977 (brand N°430468)
Natural availability :
Rhubofix® is not available in its natural state.
Isomerism :
Rhubofix® has six asymmetric carbons, offering multiple opportunities for isomerism. However, in perfumery, it is a mixture of these isomers that is used. Lilial® is a constitutional isomer of Rhubofix®. Nevertheless, their smell is very different, as it is more fruity, less floral and not aldehydic.
Synthesis precursor :
Rhubofix® is not a precursor to the synthesis of another compound of olfactory interest.
Synthesis route :
Data not available.
Stability :
Stable in perfumes and diverse functional bases
Other comments :
Present in Cassis Base 345 F® in a quite large quantity.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is not restricted for the 51th amendment



















