Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
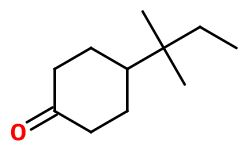
ORIVONE | ORI-1 |
Visit website
|
- | 10 grs | 92.0 - 100.0 |
General Presentation
-
CAS N° :
16587-71-6 -
EINECS number :
240-642-0 -
FEMA number :
Donnée indisponible. -
FLAVIS number :
Donnée indisponible.
-
JECFA number :
Donnée indisponible. -
Volatility :
Heart -
Price Range :
€€€€
Physico-chemical properties
-
Appearance :
Colorless liquid -
Density :
0,92 -
Refractive Index @20°C :
Donnée indisponible. -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
0,031743 mmHg @23°C -
Flash Point :
104°C (219,2°F)
-
Molecular formula :
C11H20O -
Molecular Weight :
168,28 g/mol -
Log P :
3,4 -
Fusion Point :
Donnée indisponible. -
Boiling Point :
-
Detection Threshold :
Donnée indisponible.
Chemistry & Uses
Uses in perfumery :
Orivone® is used in majority in orris root accords. It can also be used in nutty, dry woods and leather accords, to bring a dry effect and an orris root and powdery note in small quantity.
Year of discovery :
1950
Natural availability :
Orivone® is not found in nature.
Isomerism :
Orivone® is a constitutional isomer of Ethyl Linalool and so called Aldehyde C11 Lenic, but has no common facet with these two molecules.
Synthesis precursor :
Orivone® is not a precursor to the synthesis of any other molecule used in perfumery.
Synthesis route :
Orivone® can be synthesized in one step from 4-tert-amylphenol, by a selective hydrogenation reaction, catalyzed by a heterogeneous catalysor such as palladium on alumina. 4-tert-amylphenol can be taken as a starting reagent, but it can also be synthesized first by an selective alkylation reaction from phenol, on a para position, reacting with amyl chloride, with a Lewis acid catalysor.
Stability :
Stable in perfumes and in diverse functional bases.
Other comments :
Orivone® is one of the key molecule reproducing the smell of orris root.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is restricted by the 51th amendment
- Quantitative limit on the use :
-
Cat.1 Cat.2 Cat.3 Cat.4 Cat.5A B C DCat.6 0,027 % 0,0080 % 0,16 % 0,15 % 0,038 % 0,038 % 0,038 % 0,013 %Cat.5A B C DCat.6 0,038 % 0,038 % 0,038 % 0,013 %0,061 % Cat.7A BCat.8 Cat.9 Cat.10A BCat.11A BCat.12 0,24 % 0,24 %0,013 % 0,29 % 0,061 % 1,1 %0,013 % 0,013 %Cat.10A BCat.11A BCat.12 0,061 % 1,1 %0,013 % 0,013 %61 %




















