Photo credits: ScenTree SAS
Methyl Pamplemousse®
Citrus > Zesty > Grapefruit > Sandalwood > Green
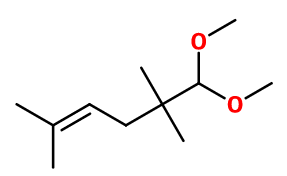
Grapefruit Acetal ; Amarocite® ; 6,6-dimethoxy-2,5,5-trimethylhex-2-ene ; 1,1-dimethox-2,2,5-trimethy-4-hexene ; 1,1-dimethoxy-2,2,5-trimethyl hex-4-ene ; 6,6-dimethoxy-2,5,5-trimethyl-2-hexene ; Methyl pomello ; Pamplerom ; Pomelocit

Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
Méthyl Pamplemousse® - 30gr | - |
Visit website
|
- | - | - | |
|
|
METHYL PAMPLEMOUSSE | AMAR-2 |
Visit website
|
- | 10 grs | - |
General Presentation
-
CAS N° :
67674-46-8 -
EINECS number :
266-885-2 -
FEMA number :
Donnée indisponible. -
FLAVIS number :
Donnée indisponible.
-
JECFA number :
Donnée indisponible. -
Volatility :
Head -
Price Range :
€€
Physico-chemical properties
-
Appearance :
Colorless liquid -
Density :
0,877 -
Refractive Index @20°C :
Donnée indisponible. -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
Donnée indisponible. -
Flash Point :
75°C (167°F)
-
Molecular formula :
C11H22O2 -
Molecular Weight :
186,29 g/mol -
Log P :
4,3 -
Fusion Point :
Donnée indisponible. -
Boiling Point :
215°C (419°F) -
Detection Threshold :
Donnée indisponible.
Chemistry & Uses
Uses in perfumery :
Methyl Pamplemousse® is used in all types of perfumes, in eau fraiche and soaps especially (for its stability), for notes of grapefruit. Gives naturalness to a grapefruit accord. Used in fougere, chypre and fruity notes to boost the head. Brings modernity to citrus fruits.
Year of discovery :
1983
Natural availability :
Methyl Pamplemousse® is not available in its natural state.
Isomerism :
Isononyl acetate is a constitutional isomer of Methyl Pamplemousse®. These two compounds do not have any olfactive similarities.
Synthesis precursor :
Methyl Pamplemousse® is not a precursor to the synthesis of another compound of olfactory interest.
Synthesis route :
Methyl Pamplemousse® is synthesized by an acetalization reaction between 2,2,5-trimethyl-4-hexenal (obtained from isobutyraldehyde and prenyl chloride) and methanol, in the presence of calcium chloride.
Stability :
Stable in perfumes and diverse functional bases, except acidic cleaners, antiperspirants and in very alkaline detergents.
Other comments :
Comparing it to other sandalwood notes as Bacdanol® and Sandalore®, Methyl Pamplemousse® has a fruity note before all, followed by a woody undernote.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is not restricted for the 51th amendment

















