Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
Grisalva® - 30gr | - |
Visit website
|
- | - | - | |
|
|
GRISALVA | GRIS-1 |
Visit website
|
- | 10 grs | - |
General Presentation
-
CAS N° :
68611-23-4 -
EINECS number :
271-889-2 -
FEMA number :
Donnée indisponible. -
FLAVIS number :
Donnée indisponible.
-
JECFA number :
Donnée indisponible. -
Volatility :
Base -
Price Range :
€€€€
Physico-chemical properties
-
Appearance :
Yellow liquid -
Density :
0,962 - 0,970 @20°C -
Refractive Index @20°C :
Donnée indisponible. -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
0,00049 mmHg @23°C -
Flash Point :
94°C (201,2°F)
-
Molecular formula :
C17H30O -
Molecular Weight :
264,41 g/mol -
Log P :
6,58 -
Fusion Point :
Donnée indisponible. -
Boiling Point :
-
Detection Threshold :
Donnée indisponible.
Chemistry & Uses
Uses in perfumery :
Used in low quantity (for cost reasons) in woody and ambery notes, in order to reproduce ambergris smell with great quality.
Year of discovery :
2007
Natural availability :
Grisalva® is exclusively resulting from a synthesis, an does not exist on its natural state.
Isomerism :
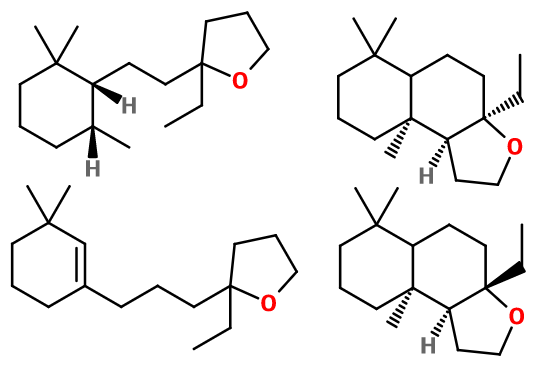
Both isomers of Grisalva® are enantiomers. They are not used separately in perfumery. The two other molecules that make up Grisalva® are very close in structure: one has a double bond on its 6-carbon cycle, while the other has none.
Synthesis precursor :
Grisalva® is not a precursor for the synthesis of another compound of olfactory interest.
Synthesis route :
Grisalva® is a mixture of four molecules, two of which are isomers. These molecules result from the reaction between prop-2-yn-1-ol and 1-(2,6,6-trimethyl-1-cyclohex-2-enyl)pentan-3-one, which is structurally very close to DihydroBeta-Ionone.
Stability :
Stable in perfumes and in diverse functional bases
Other comments :
Grisalva® takes its name from ambergris, a spermwhale rejection formerly used for perfumery.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is not restricted for the 51th amendment



















