Photo credits: ScenTree SAS
Do you sell any of the raw materials? Would you like to let our users know?
Send an email to fournisseurs@scentree.coto learn about our advertising opportunities.
Do you sell any of the raw materials? Would you like to let our users know?
Send an email to fournisseurs@scentree.coto learn about our advertising opportunities.
General Presentation
-
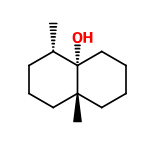
CAS N° :
23333-91-7 -
EINECS number :
Donnée indisponible. -
FEMA number :
Donnée indisponible. -
FLAVIS number :
Donnée indisponible.
-
JECFA number :
Donnée indisponible. -
Volatility :
Base -
Price Range :
€€€€
Physico chemical properties
-
Appearance :
Colorless liquid -
Density :
Donnée indisponible. -
Refractive Index @20°C :
Data not available. -
Optical rotation :
Data not available. -
Vapor pressure :
Data not available. -
Flash Point :
104°C (219,2°F)
-
Molecular formula :
C12H22O -
Molecular Weight :
182,307 g/mol -
Log P :
Donnée indisponible. -
Fusion Point :
Donnée indisponible. -
Boiling Point :
270 - 271 °C -
Detection Threshold :
(-)-geosmin : 0,0095 ppb
(+)-geosmin : 0,0780 ppb
Chemistry & Uses
Uses in perfumery :
Due to its intense strength, geosmin must be handled with extreme care in formulations. It is best used in trace amounts only. When properly dosed, it brings a moist, earthy nuance, reminiscent of patchouli or the smell of soil after rainfall.
Year of discovery :
Geosmin was first isolated in 1965 by chemists N. N. Gerber and H. A. Lechevalier from the bacterium Streptomyces griseus LP-16. Although numerous studies have been conducted to improve natural isolation yields, the synthetic route remains the preferred method of production.
Natural availability :
Geosmin is a molecule naturally produced by bacteria (Actinobacteria in soil, Cyanobacteria in water) as well as by certain species of green algae. However, in perfumery, this molecule is not available in its natural form.
Isomerism :
Geosmin exists in two enantiomeric forms: (–)-geosmin and (+)-geosmin. The laevo form is significantly more potent from an olfactory perspective. Geosmin contains four stereogenic centers, which theoretically allows for 16 possible stereoisomers, although not all of them are chemically stable.
Synthesis precursor :
Data not available.
Synthesis route :
Geosmin can be obtained through various processes, either via classical chemical synthesis or through biosynthesis, typically using bacteria. For more information, we invite you to consult the article: Enantioselective synthesis of all stereoisomers of geosmin and of biosynthetically related natural products – Org. Biomol. Chem., 2024, 22, 5748–5758.
Stability :
Data not available.
Other comments :
The name 'geosmin' comes from the Greek gê (earth) and osmé (smell).
Regulations & IFRA
Allergens :
This ingredient does not contain any allergen.
IFRA 51th :
This ingredient is not restricted for the 51th amendment

