Photo credits: ScenTree SAS
Fraistone®
Fruity > Green Fruits > Anisic > Green
Fragolane® ; Dimethyl Dioxolan ; Fraisberry® ; Ethyl 2-(2,4-dimethyl-1,3-dioxolan-2-yl) acetate ; 2,4-dimethyl-1,3-dioxolane-2-ethyl acetate ; Dimethyldioxolan ; Ethyl 2-(2,4-dimethyl-1,3-dioxolan-2-yl)acetate ; Ethyl aceto acetate PG acetal ; Ethyl acetoacetate propylene glycol ketal ; Fructone B ; Fruity ketal ; Propyl fruitat ; Strawberry ketal

Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
Fraistone | - |
Visit website
|
Synthetic Aroma Chemicals | - | - | |
|
|
FRAISTONE | FRASTN-1 |
Visit website
|
- |


|
10 grs | 97 - 100 |
General Presentation
-
CAS N° :
6290-17-1 -
EINECS number :
228-536-2 -
FEMA number :
4294 -
FLAVIS number :
06.087
-
JECFA number :
1715 -
Volatility :
Head/Heart -
Price Range :
€€
Physico-chemical properties
-
Appearance :
Colorless liquid -
Density :
1,042 -
Refractive Index @20°C :
1,422 – 1,432 -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
0,0589 mmHg @23°C -
Flash Point :
91°C (195,8°F)
-
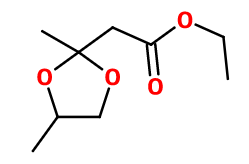
Molecular formula :
C9H16O4 -
Molecular Weight :
188,22 g/mol -
Log P :
1,5 -
Fusion Point :
-68°C (-90,4°F) -
Boiling Point :
85°C (185°F) -
Detection Threshold :
Donnée indisponible.
Chemistry & Uses
Uses in perfumery :
Fraistone® is used in fruity and floral notes of fruity rose, tuberose, jasmine, orange blossom and syringua.
Year of discovery :
1937
Natural availability :
Fraistone® is not available in its natural state.
Isomerism :
Fraistone® has two asymmetric carbons. It is however a mixture of isomers that is used in perfumery.
Synthesis precursor :
Fraistone® is not a precursor to the synthesis of another compound of olfactory interest.
Synthesis route :
Fraistone® is an acetal of Ethyl Acetoacetate (a synthesis based on formic acid and acetone in its enolic form). It is obtained by reaction between this reagent and propan-1,2-diol.
Stability :
Stable in perfumes and diverse functional bases
Other comments :
Data not available.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is not restricted for the 51th amendment



















