Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
ISOANANATE | IA-1 |
Visit website
|
- | 10 grs | 98 - 100 |
General Presentation
-
CAS N° :
68901-15-5 -
EINECS number :
272-657-3 -
FEMA number :
Donnée indisponible. -
FLAVIS number :
Donnée indisponible.
-
JECFA number :
Donnée indisponible. -
Volatility :
Heart -
Price Range :
€€
Physico-chemical properties
-
Appearance :
Colorless liquid -
Density :
1,017 -
Refractive Index @20°C :
1,460 - 1,464 -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
0,003 mmHg @25°C -
Flash Point :
101°C (213,8°F)
-
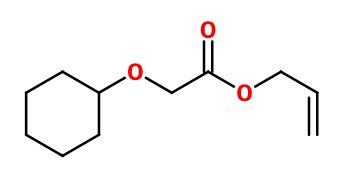
Molecular formula :
C11H18O3 -
Molecular Weight :
198,26 g/mol -
Log P :
Donnée indisponible. -
Fusion Point :
-20°C (-4°F) -
Boiling Point :
259°C (498,2°F) -
Detection Threshold :
Donnée indisponible.
Chemistry & Uses
Uses in perfumery :
Cyclogalbanate® is used in fruity notes to make them greener and give them power. Also gives a solar effect to a composition.
Year of discovery :
Data not available.
Natural availability :
Cyclogalbanate® is not available in its natural state.
Isomerism :
Cyclogalbanate® does not have any isomer used in perfumery.
Synthesis precursor :
Cyclogalbanate® is not a precursor to the synthesis of another compound of olfactory interest.
Synthesis route :
Cyclogalbanate® is synthesized by esterification of cyclohexyloxyacetic acid (prepared from phenoxyacetic acid) with allyl alcohol, catalysed by a strong acid.
Stability :
Cyclogalabante may form its corresponding acid through time under heat.
Other comments :
Cyclogalbanate is frequently compared to Allyl Amyl Glycolate for its fruity and green note. It remains much greener than the later anyway.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is not restricted for the 51th amendment

















