Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
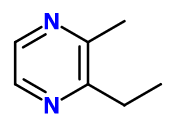
Pyrazine, 2-Ethyl-3-Methyl 1 | - |
Visit website
|
Synthetic Aroma Chemicals | - | - | |
|
|
2-ETHYL-3 METHYLPYRAZINE | 441E105000 |
Visit website
|
Synthetic Aroma Chemicals |

|
- | - |
General Presentation
-
CAS N° :
15707-23-0 -
EINECS number :
239-799-8 -
FEMA number :
3155 -
FLAVIS number :
14.006
-
JECFA number :
768 -
Volatility :
Head/Heart -
Price Range :
€€€
Physico-chemical properties
-
Appearance :
Colorless liquid -
Density :
0,987 -
Refractive Index @20°C :
1,502 - 1,505 -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
Donnée indisponible. -
Flash Point :
59°C (138,2°F)
-
Molecular formula :
C7H10N2 -
Molecular Weight :
122,17 g/mol -
Log P :
Donnée indisponible. -
Fusion Point :
Donnée indisponible. -
Boiling Point :
57°C (134,6°F) -
Detection Threshold :
2 ppm (0,0002%)
Ce qui fait de lui un composé bien moins puissant que la 2-Acétyl-pyrazine
Chemistry & Uses
Uses in perfumery :
2-ethyl-3-methyl Pyrazine si useful for reconstitutions of coffee, chocolate, hazelnut and other cooked and grilled food notes.
Year of discovery :
Data not available.
Natural availability :
2-ethyl-3-methyl Pyrazine is not available in its natural state.
Isomerism :
2-ethyl-3-methyl Pyrazine has no known isomer in perfumery.
Synthesis precursor :
2-ethyl-3-methyl Pyrazine is not a precursor to the synthesis of another compound of olfactory interest.
Synthesis route :
2-ethyl-3-methyl Pyrazine is part of a family of molecules which scent is close to peanuts as well as cooked and roasted fruits. Pyrazines are often obtained with a Gutknecht or Gastaldi condensation reaction, in both cases to condense two amines on two ketones, under the effect of an acid and oxidation. Here, one of the reagents contains the methyl and ethyl groups of the final molecule, while the other does not have any branches.
Stability :
Stable in perfumes and diverse functional bases
Other comments :
2-ethyl-3-methyl Pyrazine is more powerful than Furaneol® or 2-acetyl Pyrazine, with a burnt note in all three cases.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is not restricted for the 51th amendment



















