Photo credits: ScenTree SAS
Silvial®
Floral > Light Flowers > Aldehydes > Aquatic
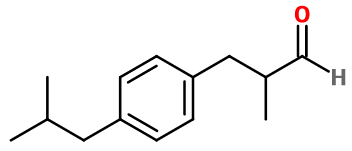
Alpha-methyl-4-(2-methylpropyl) benzenepropanal ; Para-isobutyl-alpha-methyl hydrocinnamaldehyde ; Aldéhyde para-isobutyl-alpha-methyl hydrocinnamique ; 3-(4-isobutylphenyl)-2-methylpropanal ; 3-(para-cumenyl)-2-methyl propionaldehyde ; Cyclamen homoaldehyde ; Homoaldehyde cyclamen ; 2-methyl-3-(4-(2-methyl propyl)phenyl) propanal ; Rhodial ; Suzaral

Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
Silvial® - 30gr | - |
Visit website
|
- | - | - |
General Presentation
-
CAS N° :
6658-48-6 -
EINECS number :
229-695-0 -
FEMA number :
Donnée indisponible. -
FLAVIS number :
Donnée indisponible.
-
JECFA number :
Donnée indisponible. -
Volatility :
Heart -
Price Range :
€€
Physico-chemical properties
-
Appearance :
Colorless liquid -
Density :
0,94 -
Refractive Index @20°C :
Donnée indisponible. -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
Donnée indisponible. -
Flash Point :
120°C (248°F)
-
Molecular formula :
C14H20O -
Molecular Weight :
204,31 g/mol -
Log P :
4,7 -
Fusion Point :
Donnée indisponible. -
Boiling Point :
245°C (473°F) -
Detection Threshold :
0,7 ng/l air
Chemistry & Uses
Uses in perfumery :
Silvial® is usually used to replace regulated aldehydes as Lilial® or Lyral® in lily of the valley accords for example. It brings a powerful and fresh note, with a floral identity.
Year of discovery :
Patent N°1.430.164 (FR) published on Nov.13, 1964 by Rhône-Poulenc company
Natural availability :
Sylvial® is not found in nature. Thus, it is not extracted from any plant.
Isomerism :
Silvial® has one asymmetric carbon. Nevertheless, a mixture of these two molecules is used in perfumes. Also, ortho and meta isomers of Silvial® are not used in perfumery. Eventually, Lilial® is a positional isomer of Silvial®. Its smell is fresher and fruitier, and less aqueous.
Synthesis precursor :
Reacting with Methyl Anthranilate, Silvial® can help synthesizing its Schiff base, although it is very poorly used.
Synthesis route :
As it has a structure close to Lilial® and Cyclamen Aldehyde®, the synthesis of Silvial® can be made in two different ways. The first one consists in a condensation of two aldehydes : 4-isobutylbenzaldehyde and Acetaldehyde, using an excess of the first reagent, and adding the second one slowly, to prevent autocondensation of Acetaldehyde. Then, a catalytic hydrogenation of the obtained product leads to Silvial®. The second synthetic route is a Friedel and Craft reaction involving isobutylbenzene and Methacrolein diacetate, followed by subsequent acidic hydrolysis of the intermediary product.
Stability :
Very unstable in stong acidic (detergents) and alkaline (liquid bleach) bases. Stable in other types of bases.
Other comments :
Sylvial® has a structure close to Lilial®, Cyclamen Aldehyde®, Bourgeonal™ and Florhydral®. Each of these molecules have a light flower smell, with a more marine nuance for Cyclamen Aldehyde®, greener for Bourgeonal™ and fruitier for Florhydral® for example.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is restricted by the 51th amendment
- Quantitative limit on the use :
-
Cat.1 Cat.2 Cat.3 Cat.4 Cat.5A B C DCat.6 0,08 % 0,053 % 0,8 % 0,99 % 0,25 % 0,25 % 0,25 % 0,083 %Cat.5A B C DCat.6 0,25 % 0,25 % 0,25 % 0,083 %0,08 % Cat.7A BCat.8 Cat.9 Cat.10A BCat.11A BCat.12 0,72 % 0,72 %0,083 % 1,9 % 1,9 % 5,4 %0,083 % 0,083 %Cat.10A BCat.11A BCat.12 1,9 % 5,4 %0,083 % 0,083 %No Restriction




















