Photo credits: ScenTree SAS

Photo credits: ScenTree SAS
| Company | Ingredient Name | ID | Comments | Naturality | Certifications | MOQ | Purity |
|---|---|---|---|---|---|---|---|
|
|
DERSANTOL™ | 981200 |
Visit website
|
Molecules |


|
- | - |
|
|
Sandalore - 30 Gr | - |
Visit website
|
- | - | - | |
|
|
SANDALSOFT | SANDA-2 |
Visit website
|
- | 10 grs | - |
General Presentation
-
CAS N° :
65113-99-7 -
EINECS number :
265-453-0 -
FEMA number :
Donnée indisponible. -
FLAVIS number :
Donnée indisponible.
-
JECFA number :
Donnée indisponible. -
Volatility :
Base -
Price Range :
€€
Physico-chemical properties
-
Appearance :
Colorless viscous liquid -
Density :
0,898 -
Refractive Index @20°C :
Donnée indisponible. -
Optical rotation :
Donnée indisponible. -
Vapor pressure :
Donnée indisponible. -
Flash Point :
95°C (203°F)
-
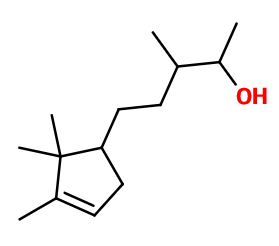
Molecular formula :
C14H26O -
Molecular Weight :
210,36 g/mol -
Log P :
4,7 -
Fusion Point :
< -50°C (< -58°F) -
Boiling Point :
275°C (527°F) -
Detection Threshold :
Donnée indisponible.
Chemistry & Uses
Uses in perfumery :
Sandalore® is used in sandalwood reproductions, woody notes for bringing volume and trail. Used for a milky and creamy effect in sandalwood notes. To be combined with other sandalwood notes such as Sandela®, Bacdanol® or Polysantol®. Not to be put in candle base. Tends to flatten accords.
Year of discovery :
1976
Natural availability :
Sandalore® is not available in its natural state.
Isomerism :
The isomer of Sandalore® formed during the synthesis has a ramified alcohol function, while Sandalore® has it on its main carbon chain. Sandalore® has two asymmetric carbons. It is therefore a mixture of isomers that is used even when Sandalore® is isolated. Adoxal® is a constitutional isomer of Sandalore®. Its smell is however radically different, as it is marine and aldehydic.
Synthesis precursor :
Sandalore® is not a precursor to the synthesis of another compound of olfactory interest.
Synthesis route :
Sandalore® is synthesized by a condensation of Campholenal with Methyl Ethyl Ketone. Then, two compounds are obtained and catalytically hydrogenated to ensure a selectivity of this hydrogenation (the alcene function of the ring must not be hydrogenated). A mixture of two hydrogenated molecules is obtained. These two isomers can be separated by a fractional distillation, or used together, as their smell is similar. The term Sandalore® refers to only one of these two molecules.
Stability :
Unstable in acid cleaners and in very alkaline products.
Labelling
Allergens :
This ingredient does not contain any allergen.
IFRA
IFRA 51th :
This ingredient is restricted by the 51th amendment
- Quantitative limit on the use :
-
Cat.1 Cat.2 Cat.3 Cat.4 Cat.5A B C DCat.6 0,21 % 0,062 % 1,2 % 1,2 % 0,29 % 0,29 % 0,29 % 0,29 %Cat.5A B C DCat.6 0,29 % 0,29 % 0,29 % 0,29 %0,68 % Cat.7A BCat.8 Cat.9 Cat.10A BCat.11A BCat.12 2,4 % 2,4 %0,12 % 2,3 % 8,1 % 8,1 %4,5 % 4,5 %Cat.10A BCat.11A BCat.12 8,1 % 8,1 %4,5 % 4,5 %No Restriction


















